THC Oxidation to CBN
CBN is the oxidative degradation product of THC. It is more stable than THC due to its conjugated structure.
![]()
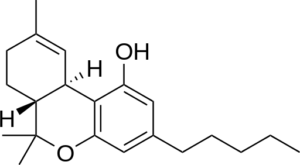
THC- Delta 9 CBN -New double bonds-Hydrogens lost here
The change is the aromatization of the top ring. 4 hydrogen atoms are lost, and two new double bonds are formed. Creating a double bond where there were hydrogens is an oxidation process, where as adding hydrogens to a double bond is a reductive process. Radical oxygen is responsible for the atmospheric degradation of THC to CBN. There are other chemical processes to synthesize CBN from THC as well.
THC Reduction to HHC

![]()
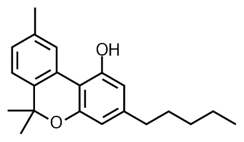
THC Delta 9 Hexahydrocannabinol (HHC)
Hexahydrocannabinol has a saturated (full hydrogen capacity, no double bonds) top ring. In this sense, it is the opposite of CBN. CBN is oxidized THC and HHC is reduced THC.
THC Delta 9 Isomerization to Delta 8 and Delta 10
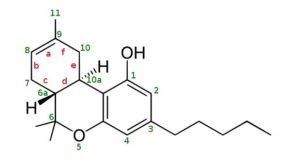
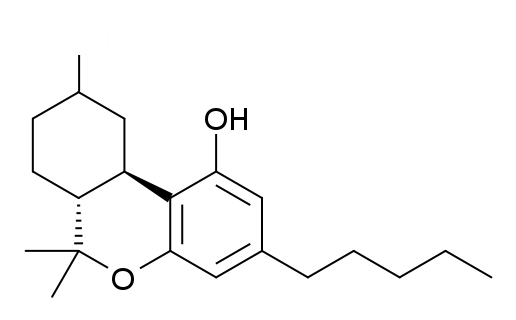
THC Delta 8, Double Bond is between 8 and 9
Delta-9, the most common and the natural form of THC, is actually the least stable.
Delta 8 has the double bond in a more thermodynamically stable position, and delta 10 even more so due to its conjugation with the bottom aromatic ring.